Potassium Hydroxide in Modern Industry: Engineering Principles, Membrane-Cell Production, and Materials Science Applications for High-Purity KOH
Release time: 2025-11-11
Potassium hydroxide (KOH) is a fundamental alkali used throughout chemical manufacturing, electrochemistry, materials processing, semiconductor fabrication, and advanced surface treatment. However, in modern industry, KOH is not merely a commodity alkali: its purity, impurity profile, production route, particle formation mechanism, and electrochemical behavior directly determine downstream performance in batteries, electroplating, fertilizers, surfactant synthesis, and silicon etching.
This article provides a high-engineering-depth, materials-science-oriented SEO analysis of KOH as an industrial reagent. It covers membrane-cell technology, electrochemical principles, impurity impacts, microstructure effects, and performance-critical applications. Tree Chem is referenced as an example of controlled-purity membrane-grade KOH manufacturing for global buyers.

High-purity potassium hydroxide (KOH) plays a critical role in membrane-cell production, electrochemical systems, semiconductor etching, and advanced materials processing. This article explains the engineering mechanisms, impurity control strategies, and performance requirements that define modern industrial-grade KOH.
1. Engineering Principles of KOH Manufacturing: Why Membrane-Grade Matters
1.1 Modern Chlor-Alkali Engineering Framework
KOH is produced by the electrolytic oxidation of aqueous potassium chloride (KCl). Two industrial technologies remain relevant:
- Ion-exchange membrane technology (IEM) – modern, clean, high-purity
- Diaphragm technology – older, chloride-contaminated, low-purity
From an engineering perspective, the membrane process has become the global standard because it enables controlled impurity migration, high current efficiency, reduced parasitic reactions, and lower downstream purification load.
1.2 Electrochemical Basis of Membrane-Cell Production
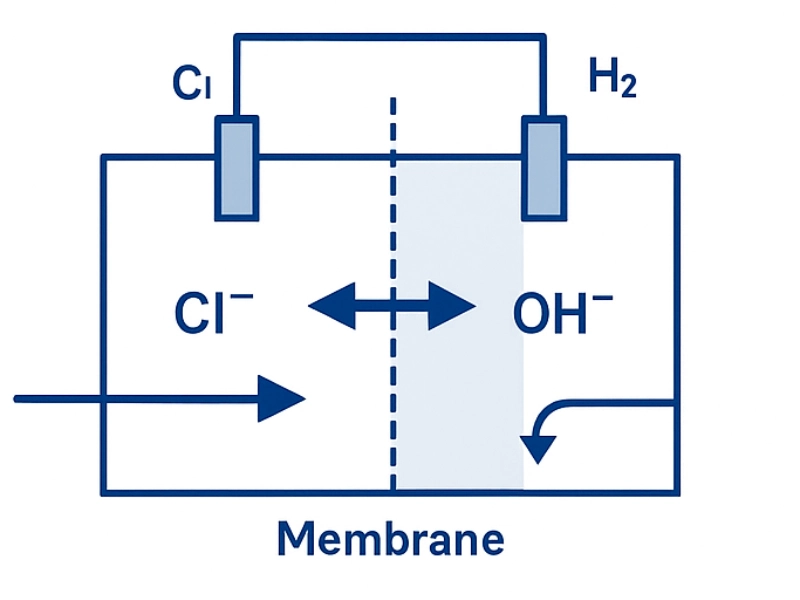
In an IEM system, an anion-blocking membrane separates anode and cathode chambers. Its polymer matrix (typically perfluorinated sulfonic acid/ether structures) allows selective cation migration while suppressing chloride crossover.
Anode Reaction
2Cl− → Cl2(g)+2e−
Cathode Reaction
2H2O+2e− → 2OH−+H2(g)
Migration Mechanism
Only K⁺ ions migrate through the membrane. OH⁻ remains in the catholyte; Cl⁻ is confined to the anolyte. This isolation is the basis for achieving low chloride (<0.005–0.02%) KOH solutions.
Engineering Performance Advantages
- Lower cell voltage
- Reduced back-diffusion of Cl⁻
- Well-defined impurity profile
- Enhanced energy efficiency
- High operational stability
Tree Chem employs membrane-cell systems designed around low-resistance membranes and optimized brine purification, enabling production of 90% / 95% flakes and high-purity liquid KOH with strict control over Fe, Ni, silica, sodium, and chloride levels.
1.3 Evaporation, Concentration, and Solidification Engineering
After electrolysis, 30–32% KOH solution is concentrated via multi-effect evaporators. Engineering design must address:
- Scaling tendency (carbonates, silicates)
- Fe/Ni pickup from equipment
- Local supersaturation leading to solid inclusions
- Moisture distribution in flake/pellet formation
Solid Flake Formation (Drum Flakers)
Controlled cooling transforms molten KOH into uniform flakes. Critical parameters:
- Cooling drum surface metallurgy
- Contact time and release temperature
- Gas-phase CO₂ exclusion
- Anti-caking flow dynamics
Pellet/Prill Formation (Spray Granulation)
Used for high-flow, dust-free grades:
- Atomization pressure
- Droplet residence time
- Humidity control to prevent deliquescence
These engineering considerations directly affect bulk density, flowability, and stability, which influence global shipping and storage performance.
2. Impurity Pathways and Their Downstream Effects
High-purity KOH is defined not only by assay (90–95%) but by trace impurity patterns. Different applications exhibit different impurity sensitivities.
Below is a technical analysis of impurity sources and their effects.
2.1 Chloride (Cl⁻): Electrochemical and Corrosion Consequences
Source:
- Incomplete membrane selectivity
- Brine purification inefficiency
- Diaphragm back-mixing
Impact:
- Accelerates pitting corrosion in stainless steel (Cl⁻ drives localized anodic dissolution)
- Alters electroplating bath composition
- Disrupts semiconductor cleaning uniformity
- Increases conductivity loss in alkaline batteries due to competing anion mobility
Even small increases in Cl⁻ (from 0.02% → 0.08%) have measurable consequences on metal finishing quality.
2.2 Heavy Metals (Fe, Ni, Cu, Zn)
Source:
- Metallic wear in electrolysis cells
- Solidification equipment (Fe pickup)
- Brine contamination
Impact:
In semiconductor and solar applications, metal ions in the ppb-to-low-ppm range generate:
- Micromask defects in anisotropic Si etching
- Non-uniform surface texturing
- Increased leakage current in photovoltaic cells
- Catalytic side-reactions during organic synthesis
Heavy metals also affect battery electrolyte stability, where Fe and Cu promote parasitic hydrogen evolution.

2.3 Silica and Insoluble Particulates
Even 5–20 ppm silica or insolubles can cause:
Residual particulate contamination in pharmaceutical production
Surface scattering in optical material processing
Roughness formation during Si etching
Nozzle/pipe blockage in high-precision dosing systems
Tree Chem uses controlled filtration and solution polishing steps to limit insoluble residues and fine particulates.
2.4 Sodium as a Process-Critical Contaminant
Na⁺ migrates into KOH if brine purification or membrane performance is suboptimal. In high-performance applications:
- Na⁺ reduces ionic conductivity compared with K⁺
- Alters electrochemical behavior in Ni–MH batteries
- Changes soap/surfactant solubilization dynamics
- Interferes with precision semiconductor processing
For advanced materials processing, Na⁺ must be minimized to prevent charge-carrier contamination.
3. Materials Science Applications: Mechanistic Analysis
This section illustrates how potassium hydroxide performs in specific high-technology processes, focusing on materials behavior rather than generic application lists.
3.1 KOH in Alkaline Batteries: Ionic Mobility and Electrochemical Stability
Why KOH instead of NaOH?
- K⁺ has higher ionic mobility (lower activation energy of migration)
- Better low-temperature conductivity
- Superior stability of zincate species
- Faster diffusion kinetics in porous electrodes
Critical Purity Parameters
- Low carbonates to prevent Zn precipitation
- Low Fe/Cu to avoid parasitic hydrogen evolution
- Uniform conductivity to maintain consistent discharge profiles
In high-drain alkaline cells, electrolyte consistency directly influences polarization behavior and cycle efficiency.
3.2 KOH in Electroplating: Cathodic Polarization and Surface Uniformity
Potassium hydroxide is widely used for substrate degreasing and metal activation.
From a materials perspective:
- OH⁻ promotes saponification of organic contaminants
- K⁺ minimizes salt precipitation vs Na⁺, keeping bath conductivity stable
- Trace metal impurities can deposit into coating defects via uncontrolled reduction
In nickel plating, KOH concentration affects cathodic overpotential, modifying grain size and deposit morphology.

High-purity KOH reduces:
- Pitting
- Micro-voids
- Localized discoloration
Making it essential for precision metal finishing operations.
3.3 KOH in Silicon Etching: Crystallographic Selectivity and Defect Control
KOH is a standard etchant for silicon wafers in MEMS, PV, and semiconductor processes.
Mechanism
Etching proceeds via nucleophilic attack of OH⁻ on Si–Si backbonds, generating soluble silicates.
Crystallographic Behavior
- {100} planes etch fastest
- {111} planes etch slowest
- Resulting in anisotropic pits or V-grooves
Why impurities matter
- Fe, Cu, Ni form catalytic micromasks → roughness
- Silica particles cause hillock formation
- Sodium contamination influences surface charge and modifies etch rate anisotropy
- Cl⁻ destabilizes surface oxide passivation
In PV manufacturing, texture uniformity directly affects optical absorption and conversion efficiency.
Purity Requirements
- Low metals (<ppm range)
- Low particulates
- Low Na⁺
- Consistent OH⁻ activity
Tree Chem’s high-purity membrane KOH is used in these processes due to stable impurity fingerprints.
4. KOH in Chemical Synthesis and Surfactant Manufacturing
In organic synthesis, KOH drives dehydrohalogenation, condensation, transesterification, amination, and polymer functionalization.
Why KOH is chemically superior
- Stronger base than NaOH (higher activity)
- More effective in alcoholysis of triglycerides
- Better solubilization of fatty acids
- More compatible with high-viscosity organic phases
In Surfactants & Quaternary Ammonium Compounds
KOH promotes:
- Efficient fatty acid neutralization
- Formation of potassium soaps with higher solubility
- Controlled amine/quaternary ammonium synthesis
Here, heavy-metal and chloride contamination cause color formation, oxidized byproducts, and slower kinetics.
5. Engineering Considerations for Storage, Handling & Packaging
High-purity KOH requires specialized engineering controls.
5.1 Hygroscopic and Carbonation Behavior
KOH rapidly absorbs moisture and CO₂, forming carbonates that:
- Change alkalinity
- Alter conductivity
- Reduce surfactant performance
- Disrupt Si etching kinetics
5.2 Packaging Engineering
High-performance packaging requires:
- Moisture-barrier liners
- Anti-corrosive drum coatings
- CO₂-reduced headspace
- Temperature-stable containers
5.3 Bulk Logistics
For global buyers:
- Flake/pellet stability under temperature cycling
- Dust suppression
- Flowability over long-distance shipping
Tree Chem applies controlled flaking and sealed-barrier packaging to maintain KOH purity during international transport.
6. How to Choose a High-Purity KOH Supplier (Engineering Checklist)
Below is a technical, engineering-driven checklist for buyers searching for a potassium hydroxide supplier, potassium hydroxide manufacturer, or high-purity KOH provider.
1. Production Route
Insist on membrane-cell KOH for stable impurity profiles.
2. Brine Purification Quality
Modern brine systems eliminate Ca, Mg, Ba, and heavy metals—key to long-term batch consistency.
3. Impurity Fingerprint
Not just assay: the ppm/ppb pattern matters.
4. Carbonate Control
Essential for battery and semiconductor applications.
5. Particle Formation Engineering
Uniform flakes/pellets indicate controlled thermal granulation.
6. Filtration and Polishing
Necessary for low-insoluble grades.
7. Batch-to-Batch Repeatability
Critical for electroplating, PV, and chemical synthesis.
8. Packaging System
Moisture barrier + CO₂ exclusion = stability during shipping.
9. Documentation & Traceability
COAs must include heavy metals, sodium, chloride, carbonate, insolubles.
10. Application Support
Suppliers should understand the engineering requirements of batteries, semiconductors, MEMS, and plating.
Tree Chem acts as a membrane-grade potassium hydroxide manufacturer offering engineered impurity control and multi-grade availability (90%, 95%, and high-purity liquid KOH).
Conclusion
Modern potassium hydroxide is not a commodity chemical but a precision-critical engineered alkali whose production route, impurity profile, and microstructure determine performance across high-tech industries. Membrane-cell technology has made low-chloride, low-metal KOH accessible for battery manufacturers, semiconductor etching lines, electroplating facilities, and advanced chemical synthesis plants. The technical relationship between KOH purity and downstream reaction kinetics, electrochemical stability, and materials performance underscores the need for qualified, engineering-driven suppliers.
For industrial buyers evaluating global potassium hydroxide suppliers, the key lies in analyzing production technology, impurity pathways, engineering controls, and batch consistency, rather than focusing only on assay. Tree Chem demonstrates the type of controlled manufacturing, membrane-cell purity, and material-grade performance required in today’s advanced industries.










